
As the COVID-19 pandemic continues, public health authorities around the world are facing a significant challenge: ensuring a large enough supply of reagents to scale-up diagnostic testing. Researchers at Canada's Michael Smith Genome Sciences Centre at BC Cancer (GSC), the University of British Columbia (UBC) and the BC Centre for Disease Control (BCCDC) are working to develop new non-proprietary reagents to support current and future testing in British Columbia. Dr. Martin Hirst, Senior Scientist at the GSC and Associate Director of UBC’s Michael Smith Laboratory (MSL), is leading this project with support from Genome BC through its Rapid Response Funding for COVID-19 Research program.
How are people tested for COVID-19?
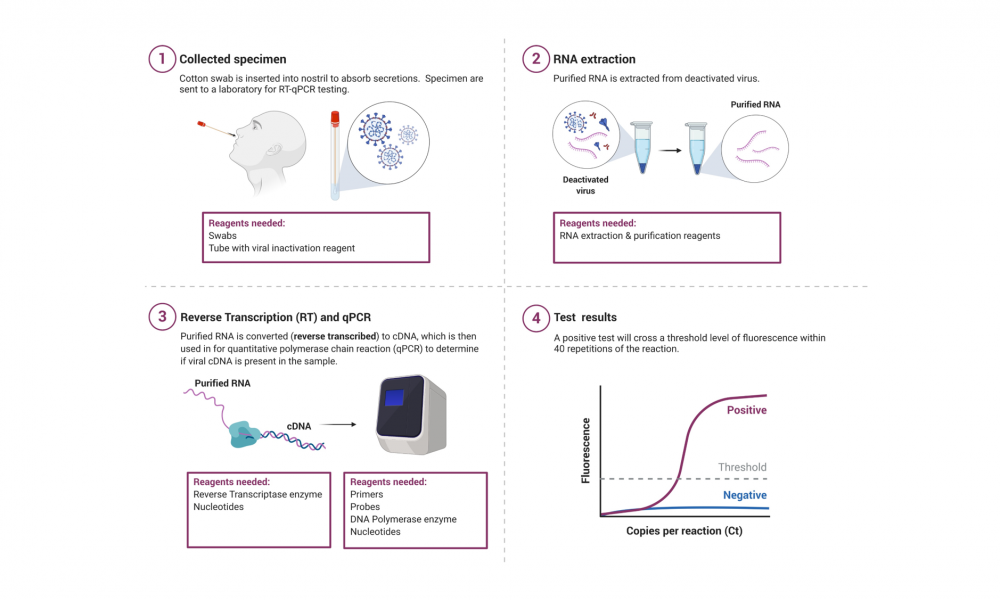
There are a couple ways. One is to test for antibodies produced by the patient in response to infection by the virus. This is useful in retrospect, to learn more about how and where populations of people were infected. The other is to test for the presence of the virus itself, in order to determine if someone is currently infected. We’re focused on the latter, using a method called reverse transcription, quantitative polymerase chain reaction (RT–qPCR) to test for SARS-CoV-2 from nose, throat or saliva samples.
How does RT-qPCR work?
Polymerase chain reaction (PCR) is a technique that causes a very small well-defined segment of DNA to be amplified, or multiplied many hundreds of thousands of times so that there is enough of it to be detected and analyzed (polymerase is an enzyme that synthesizes long chains of nucleic acids). However, viruses such as SARS-CoV-2 do not contain DNA, but RNA and “reverse transcription” is the process used to convert RNA into DNA, which has to happen for PCR to work. The amount of viral DNA is then “quantified” using chemicals that fluoresce more brightly with the presence of more DNA. If the virus is present in a sample from a nose or throat swab, for example, this method will first convert its RNA to DNA and then amplify it to the point that it becomes detectable.
What problem with testing are you aiming to address?
COVID-19 testing in B.C. involves the extraction of RNA from biospecimens using proprietary platforms, and diagnostic testing using a laboratory developed RT-qPCR test with other proprietary reagents. These proprietary reagents are under increasing global demand; extraction kits and probes are back-ordered, and contaminated testing reagents have been received stemming from an inability of companies to keep up with good manufacturing practices. We need to develop the capacity to develop reagents quickly and at the scale needed to support rapid diagnostic testing on an ongoing basis, until this current pandemic subsides and if or when we face another one like it.
How is your research supporting more testing for COVID-19?
We are using the infrastructure at the GSC, UBC and BCCDC to develop scalable and automated non-proprietary extraction and RT-qPCR workflows for the development of stable reagent supply chains. We are producing reagent formulas and automated workflows, shared initially between the UBC and BCCDC sites and, following qualification, to testing sites in B.C. and across Canada. We are working with STEMCELL Technologies to formulate and produce reagents at scale and documenting our automated workflows under Quality Management Systems to provide as turn-key solutions to testing sites in B.C. and beyond.
Where is this research happening?
Our research is being carried out at sites that are uniquely positioned to respond to this testing crisis with world class molecular diagnostic capability, and the appropriate technical and scientific expertise. Specifically, the work will be carried out at MSL and the Life Sciences Centre (LSC) at UBC, the GSC at BC Cancer and Public Health Lab at BCCDC. MSL and GSC both have equipment for biochemical purification and for benchmarking RT-qPCR assays. The LSC has the required molecular biology tools to test enzyme activities and an ABI oligonucleotide synthesis instrument and processing equipment for probe manufacture. BCCDC’s Public Health Lab has access to the samples and the equipment to carry out testing and the BCCDC has the mandate to work closely with other laboratories, clinicians, Medical Health Officers, epidemiologists, the Provincial Health Officer and others to support expanding COVID-19 testing to a greater population.
How will this project impact the current COVID-19 pandemic?
Our research is ensuring a secure supply of laboratory reagents needed in B.C. to maintain and expand its capacity for COVID-19 testing. In the medium term, these reagents will be further validated and production will be scaled-up to meet accelerating testing demands in the province. The establishment of molecular resources, protocols and formulations to support large scale RT-qPCR viral testing will also ensure that Canada is prepared to meet future pandemics, particularly in the case of any disrupted global supply chains. Ultimately, a stable supply of reagents, produced locally, will ensure testing can meet demands, resulting in enhanced contact tracing and isolation that will ultimately limit transmission of SARS-CoV-2 and help to manage the spread of COVID-19.
Who is participating in this research?
This project is a collaborative effort between six B.C. labs, including, at the GSC, Drs. Robert Holt, Gregg Morin and Martin Hirst; at MSL, Dr. Thibault Mayor; and at LSI, Drs. Sheila Teves and Ivan Sadowski. A few GSC scientists and staff deserve special mention, including the leadership of Dr. Marco Marra and Dr. Steven Jones. Dr. Eric Yung in the Holt lab designed and sourced the four essential enzymes in the assay. Dr. Grace Cheng in the G. Morin lab has been purifying the Taq polymerase and RNasin proteins. Dr. Robin Coope, Dr. Yongjun Zhao, Dr. Simon Merhu, Stephen Pleasance, Pawan Pandoh and Duane Smailus are developing custom automated RNA extraction workflows. Miruna Bala, Olivia Griffith, Paul Lythgo, Dr. Andy Mungall, Jessica Nelson, Robyn Roscoe, Kevin Sauvé, Dr. Sherry Wang, Cavin Wong and George Yang are providing critical assistance with administration and logistics. Michelle Moksa, Marcus Wong, Qi Cao and Edmund Su are GSC scientists in the Hirst lab at the MSL who have been working on qualification of alternate qRT-PCR reagents. These individuals have set aside their own research and have spent a significant amount of time in the lab to move this project forward.